Most vaccines prepared to fight deadly diseases contain adjuvants that make their reaction stronger by releasing a more robust immune response. Distinguishing potential adjuvants just got simpler, based on a methodology portrayed by researchers at Kyoto University’s Institute for Integrated Cell-Material Sciences (iCeMS)
The group of scientists and researchers in Japan reported that they have found a particle that, when added to an immunization, reinforces the safe reaction similar to that of an ordinarily utilized adjuvant. Immunization adjuvants are a fundamental piece of clinically utilized antigens used to trigger antibodies.
“Adjuvants generate a robust and long-lasting immune response, but the ones currently in use, like aluminium salts and oil-in-water emulsions, were developed in the 1920s, and we don’t precisely understand how they work, which is why they are often called immunologists’ dirty little secret,” exclaimed iCeMS chemical biologist Motonari Uesugi, who led the study.
How do vaccines work?
A vaccine works by preparing the immunity framework to perceive and battle microorganisms, either infections or microscopic organisms. To do this, the specific microorganism should be acquainted with the body to trigger an insusceptible reaction.
These microorganisms used in the entire process are called antigens. By infusing these antigens into the body, the resistant framework can securely figure out how to remember them as threatening intruders, produce antibodies, and prepare the body for what’s to come. If the microbes or infections return, the safe framework will perceive the antigens promptly and protect our body from the specific ailment’s effects.
How was such strenuous research conducted?
The new adjuvant was found by screening a library of 8,000 little atoms for their capacity to self-collect. Sub-atomic self-gathering is simply the unconstrained association of particles through non-electron-sharing bonds. This is a notable idea in material science that is additionally utilized by living life forms to perform complex organic capacities.
“We hypothesized that structures that come together through molecular self-assembly might mimic structures in pathogens, like viruses, stimulating a similar immune response,” says Uesugi.

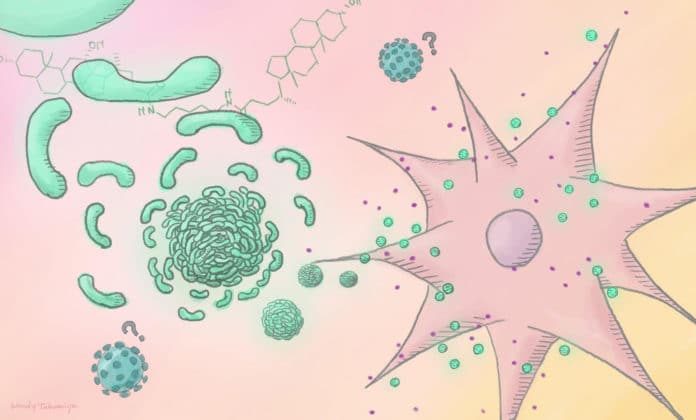
The group discovered 116 particles that can self-collect, and screened them for the capacity to build interleukin-6 articulation by macrophages. Macrophages are resistant cells that recognize and “eat up” microorganisms coursing in the body. They additionally discharge proteins, for example, interleukin-6, that enact other safe cells.
The examination prompted the disclosure of a particle called cholicamide. This atom self-collected to shape an infection impersonating structure that is inundated by macrophages and comparable safe cells. The structures are moved into particular vacuoles to consolidate with a particular receptor called cost like receptor 7, which starts an elevated invulnerable reaction. In particular, it prompts the arrival of safe, stimulating signals like interleukin-6.
What are the future prospects of the research?
Further examinations and correlations exhibited that cholicamide was similar in intensity to the adjuvant Alum in instigating an insusceptible reaction when added to a flu immunization given to mice.
Investigations are expected to decide how cholicamide impersonates the single RNA strands of infections to initiate cost like receptor 7. The scientists likewise need to see how cholicamide ties to the receptor to clarify this connection’s impacts.
Uesugi further added, “Our study, to the best of our knowledge, is the first report of using a small molecule library for vaccine adjuvant discovery. We hope the new approach paves the way for discovering and designing self-assembling small molecule adjuvants against pathogens, including emerging viruses.
Further Reading: